A New Path Toward Disease Modification
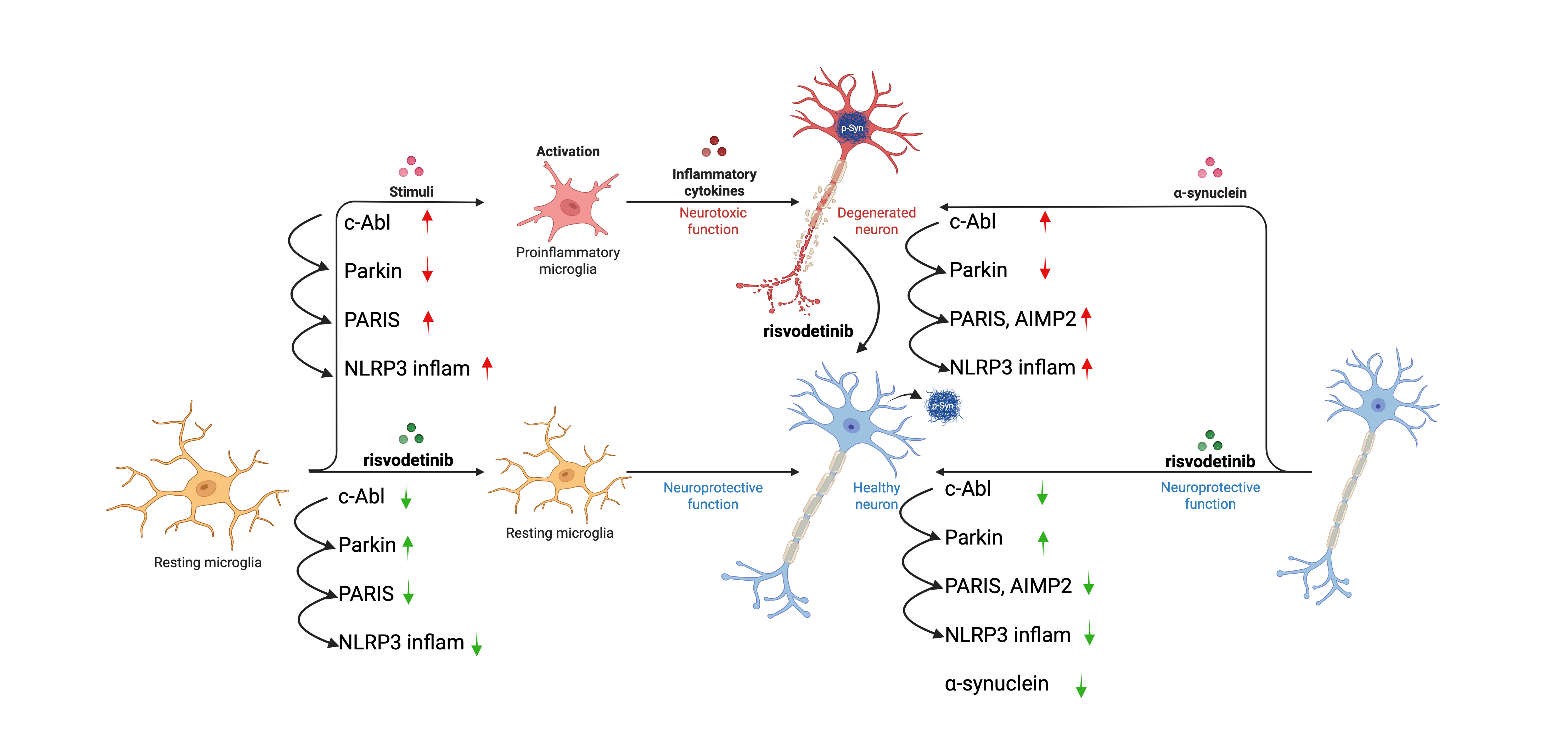
Unlike traditional treatments that only replace lost dopamine, Risvodetinib aims to target the cause of neuronal death itself. Recent data from our Phase 2 Trial has confirmed that c-Abl is likely a master regulator of neurodegeneration in Parkinson’s disease. c-Abl activation in neurons and in microglia, what might be viewed as the ‘immune cell of the brain’, drives neurodegeneration by inducing cell death by a variety of mechanisms through a signaling cascade present in both neurons and microglia. Risvodetinib restores the brain’s natural defense systems, thus c-Abl inhibition looks like it may reverse the progression of Parkinson’s disease. We believe this represents a significant step toward modifying human Parkinson’s disease. Risvodetinib blocks both neurodegenerative cell death pathways and neurodegenerative inflammatory pathways, as illustrated in the figure below.
Understanding the Root Cause
In Parkinson’s disease, nerve cells, or neurons, that control movement gradually die because a structural protein alpha-synuclein becomes misfolded and internalized by the affected neurons.
Once internalized, these misfolded proteins activate a stress-response enzyme called c-Abl. Once triggered, c-Abl modifies alpha-synuclein to create the toxic, disease-causing form of the protein. This, in turn, triggers programs of cell death through several mechanisms in neurons.
As a result, mitochondria falter, damaged proteins pile up, and a neuron slowly deteriorates leading to the classic motor symptoms of Parkinson’s disease. These processes are represented in the figure below:

The c-Abl Connection
ABLi’s research has shown that c-Abl acts as the master regulator for this destructive process.
When c-Abl is active:
- Toxic alpha-synuclein accumulates in the neuron and can spread to neighboring neurons through tunnels formed between them.
- Parkin, the protein that maintains mitochondrial function and clears toxic proteins, is turned off.
- Cellular repair systems collapse, driving nerve-cell death through several mechanisms.
How Risvodetinib Works
Risvodetinib is designed to block c-Abl activity inside the neuron, halting this cycle of destruction.
When c-Abl is inhibited:
- Toxic alpha-synuclein is destroyed within the neuron intrinsic mechanisms like the ubiquitin/proteosome system and autophagy/lysosomal clearance mechanisms.
- Parkin function is restored, allowing the cell to clear toxic alpha-synuclein and restore normal mitochondrial function.
- Nerve cells begin to recover, leading to improvements in both motor and non-motor functions in preclinical studies.